SOLUTIONS
**Chapter Objectives:
- Distinguish between homogeneous and heterogeneous mixtures.
- Define and be able to identify in a chemical system: solution, solute, solvent
- List three different solute-solvent combinations.
- Define and provide examples of solubility.
- Read a solubility curve.
- Compare and contrast the terms saturated, unsaturated, and supersaturated.
- Describe the steps of solution formation.
- Define heat of solution. Explain why some heats of solution formation are exothermic, and some endothermic.
- From the temperature change that occurs when a given solution is made, determine if the dissolving process was endothermic or exothermic.
- List and explain factors that affect the rate of dissolving.
- List and explain the factors that affect solubility.
- Calculate molarity (M) & use the definition of molarity to solve for amount of solute (in grams or moles) or solve for volume of solution.
- Describe how to make a given amount of solution of known molarity from both a solid solute and a concentrated stock solution.
- Solve dilution problems (McVc = MdVd)
- Calculate % by mass for a given solution, or the mass of solute or mass of solvent given % mass of the solution.
- Define concentrated, dilute, miscible, immiscible.
- Predict the solubility of a solute in a given solvent based on their polarities. (Like dissolves like.)
- Know that colligative properties are properties that depend on the concentration of particles in solution but not on the nature of the particles.
- List three colligative properties, and explain how the presence of the solute impacts the physical property of the solvent.
- Describe the effects of a nonvolatile solute on the freezing point and the boiling point of water.
- Calculate freezing-point depression, boiling point elevation, and solution molality of nonelectrolyte solutions.
- Given freezing point and boiling point data, calculate the molar mass of the solute.
- Compare the properties of suspensions, colloids, and solutions.
Introduction to Solutions
(basic vocabulary, solubility)
(Content in this video will be assessed during the week of April 27, 2020.)
Sample Questions (measure understanding of Solution basics)
Additional Neat Stuff (Optional!):
Additional Video: Saturated, Unsaturated, Supersaturated Solutions & Solubility Curves
Solubility Curve Worksheet - Answers Included
*This worksheet practices Interpreting a solubility curve -
consult the first two pages only (problems only pages 3-5 do not apply to us this unit)
Heterogeneous vs.
Homogeneous Mixtures
(Solutions vs. Colloids vs. Suspensions)
(Content in this video will be assessed during the week of April 27, 2020.)
Homogeneous Mixtures
(Solutions vs. Colloids vs. Suspensions)
(Content in this video will be assessed during the week of April 27, 2020.)
Solution Formation
(Content in this video will be assessed during the week of April 27, 2020.)
Factors that affect Rate of Soln Formation & Factors that affect Solubility
(Content in this video will be assessed during the week of April 27, 2020.)
(Content in this video will be assessed during the week of April 27, 2020.)
Sample Questions
(measure understanding of factors that impact rate of solution formation
and solubility)
SOLUTION CONCENTRATION EXPRESSIONS
(% Comp, M, m)
(Content in this video will be assessed during the week of May 4, 2020.)
(measure understanding of factors that impact rate of solution formation
and solubility)
SOLUTION CONCENTRATION EXPRESSIONS
(% Comp, M, m)
(Content in this video will be assessed during the week of May 4, 2020.)
Additional Tutorial of Solution Concentration Problem Solving
(Content in this video will be assessed during the week of May 4, 2020.)
Sample Questions
(measure understanding of Solution Concentration Expressions)
Sample Problems
*ignore problem #9 - deals with colligative property (next week's topic)
COLLIGATIVE PROPERTIES
*Introduction to Vapor Pressure Lowering, Boiling Point Elevation,
and Freezing Point Depression
(Content in this video will be assessed during the week of May 11, 2020.)
(measure understanding of Solution Concentration Expressions)
Sample Problems
*ignore problem #9 - deals with colligative property (next week's topic)
COLLIGATIVE PROPERTIES
*Introduction to Vapor Pressure Lowering, Boiling Point Elevation,
and Freezing Point Depression
(Content in this video will be assessed during the week of May 11, 2020.)
Additional Neat Stuff (not required)
Additional Tutorial Videos: Colligative Properties of Solutions
Colligative Properties
Electrolytes
(Content in this video will be assessed the week of May 11th.)
Electrolytes (continued)
Dissociation Equations, Van't Hoff Factor
(Content in this video will be assessed the week of May 11th.)
Sample Questions (measure understanding of dissociation, electrolytes)
Debye Huckel Theory (addressed during week of May 11. 2020)
Debye Huckel Theory (addressed during week of May 11. 2020)
Electrolytes (continued)
Acids - Weak vs. Strong
(Content in this video will be assessed the week of May 11th.)
Colligative Property Problem Solving
(Content in these videos will be assessed during the week of May 11, 2020.)
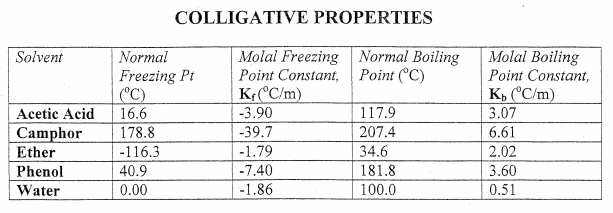
Problem Worksheet used in Colligative Property Problem Solving Part II
(nice example problems with work shown)
(nice example problems with work shown)
Problem Worksheet used in Colligative Property Problem Solving - Electrolyte vs. Nonelectrolyte
**Only Problem #3 and Problem #4 on this worksheet
**Only Problem #3 and Problem #4 on this worksheet
Sample Questions (measure understanding of ionization, dissociation, electrolytes, strong and weak acids, freezing point depression, boiling point elevation)
REVIEW MATERIALS (of Soln Unit):
1. Practice Test 1
Practice Test 1 Key
2. Practice Test 2
Practice Test 2 Key
3. Concentration and Colligative Property Problems (work and key included)
4. Theory Questions / Chapter Objectives