"Mini" Unit - Chpt 14 & 19
*Specific Solute Particles (dissociation and ionization)
Driving Forces of Chemical Reactions
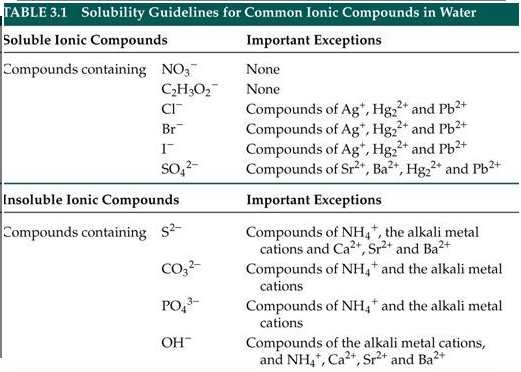
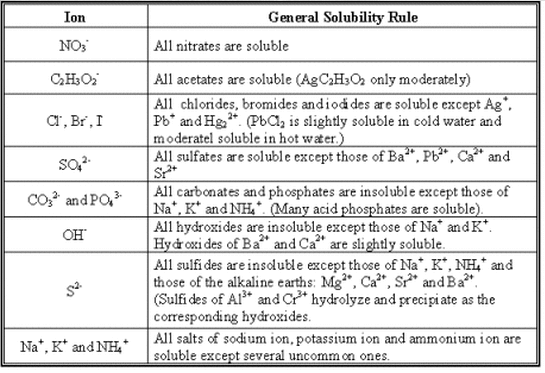
*Solubility Rules
*Electrolytes
Oxidation-Reduction Reactions
*Ionic and Net Ionic Equations
*2020 Topics
*Specific Solute Particles (dissociation and ionization)
Driving Forces of Chemical Reactions
*Solubility Rules
*Electrolytes
Oxidation-Reduction Reactions
*Ionic and Net Ionic Equations
*2020 Topics
Chapter Objectives:
- Know the four driving forces of chemical reactions.
- Given a chemical equation, be able to identify the driving force.
- Identify electrolytes and nonelectrolytes from their formulas.
- Applying a solubility table, predict if a given ionic solute will be soluble or insoluble in water.
- Distinguish between dissociation and ionization of a solute and write equations describing these processes.
- Differentiate between weak and strong electrolytes.
- Know that colligative properties are properties that depend on the concentration of particles in solution but not on the nature of the particles.
- Given a selection of solute/solvent systems, determine which will display the greatest/least freezing point depression or boiling point elevation.
- Determine how many particles a solute will form when it dissolves. Write appropriate dissolution equations for substances such as C6H12O6, NaCl, MgCl2, AlCl3.
- Calculate freezing-point depression and boiling point elevation for electrolyte solutions.
- Complete double replacement reactions and use solubility rules to predict precipitates in double replacement reactions.
- Write complete formula equations, complete ionic equations, and net ionic equations for double replacement reactions.
- Compare and contrast oxidation and reduction.
- Identify what species is being oxidized and what species is being reduced in a given chemical reaction.
IONIC AND NET IONIC REACTIONS:
(assessed during week of June 1, 2020)
Sample Solubility Charts -
|
Sample Questions
Applying Solubility Rules & Writing Ionic and Net Ionic Equations
Key/Answers found after questions.